Difference between revisions of "Os01g0262600"
(→Function) |
|||
| Line 4: | Line 4: | ||
'''T-DNA Insertions in OsGT1 Cause a Defect in Male Gametophyte Development''' | '''T-DNA Insertions in OsGT1 Cause a Defect in Male Gametophyte Development''' | ||
| + | |||
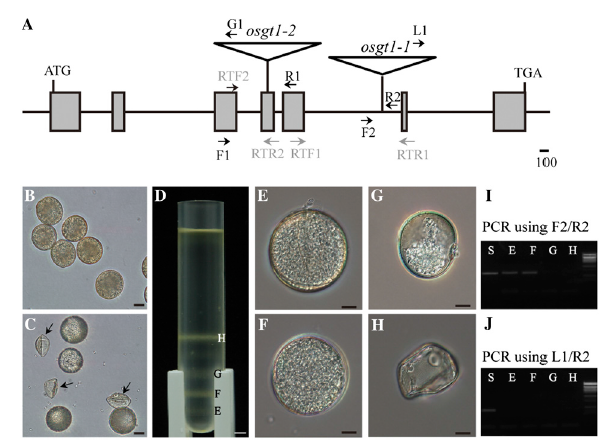
We previously generated transfer DNA (T-DNA)-tagged lines of japonica rice (Jeon et al., 2000; Jeong et al., 2002) and determined flanking sequences for the insertion sites (An et al., 2003; Jeong et al., 2006). To identify genes essential for gametophyte development, we genotyped the T-DNA insertion lines; those with a segregation ratio close to 1:1:0 (wild type:heterozygote: homozygote) were selected. From 541 independentlines, we obtained eight with the segregation distortion phenotype. Here, we report detailed analyses of line 3C-00590, which carries a T-DNA insertion in the fifth intron of LOC_Os01g15780 (Fig. 1A). Because the gene encodes a protein in the glycosyltransferase family, we named it OsGT1. We found that another allele (osgt1-2) also harbors T-DNA in the fourth exon of OsGT1 (Fig.1A). This line also exhibited a 1:1:0 segregation (Table I;Supplemental Fig. S1), confirming that this phenotype is due to the T-DNA insertion to OsGT1. | We previously generated transfer DNA (T-DNA)-tagged lines of japonica rice (Jeon et al., 2000; Jeong et al., 2002) and determined flanking sequences for the insertion sites (An et al., 2003; Jeong et al., 2006). To identify genes essential for gametophyte development, we genotyped the T-DNA insertion lines; those with a segregation ratio close to 1:1:0 (wild type:heterozygote: homozygote) were selected. From 541 independentlines, we obtained eight with the segregation distortion phenotype. Here, we report detailed analyses of line 3C-00590, which carries a T-DNA insertion in the fifth intron of LOC_Os01g15780 (Fig. 1A). Because the gene encodes a protein in the glycosyltransferase family, we named it OsGT1. We found that another allele (osgt1-2) also harbors T-DNA in the fourth exon of OsGT1 (Fig.1A). This line also exhibited a 1:1:0 segregation (Table I;Supplemental Fig. S1), confirming that this phenotype is due to the T-DNA insertion to OsGT1. | ||
These segregation phenotypes suggested that the defect results from a failure in gene transfer through either the male or female gamete. To clarify the function of OsGT1 in the gametophyte, we performed reciprocal crosses between heterozygotes (OsGT1/osgt1-1) and the | These segregation phenotypes suggested that the defect results from a failure in gene transfer through either the male or female gamete. To clarify the function of OsGT1 in the gametophyte, we performed reciprocal crosses between heterozygotes (OsGT1/osgt1-1) and the | ||
| Line 9: | Line 10: | ||
'''osgt1 Grains Are Defective at the Mature Pollen Stage''' | '''osgt1 Grains Are Defective at the Mature Pollen Stage''' | ||
| + | |||
To examine further the morphological defects in male gametes of osgt1-1, we collected pollen grains from fully developed anthers and observed them with a bright field microscope. Pollen from wild-type anthers was normal: only a small fraction (nine out of 1,393) was shrunken at stage 12 (Fig. 1B; Supplemental Fig. S2). However, anthers from the heterozygote produced shrunken grains at a 49.5% frequency (915 of 1,847; Fig. 1C; Supplemental Fig. S2), indicating that OsGT1 is required for male gamete development. To determine whether those shrunken grains were caused by the T-DNA insertion, we used a Suc density gradient to separate them from normal grains (Bedinger and Edgerton, 1990; Fig. 1D). The gradient containedfour discrete bands, two lower bands that contained normal pollen with a globular shape (Fig. 1, E and F) plus two upper bands with defective grains that were shrunken or underdeveloped (Fig. 1, G and H). To see whether this defect was due to the mutation, we prepared DNA from each band and performed PCR with gene-specific primers. This resulted in amplification of the OsGT1 fragment from the two lower bands but not from the upper bands (Fig. 1I). Amplification with a gene-specific primer (R2) and a T-DNA primer (L1) did not generate bands from any of the four samples, indicating that normal pollen in the lower bands did not carry T-DNA and that abnormal pollen in the upper bands lacked genomic DNA because they were dead (Fig. 1J). | To examine further the morphological defects in male gametes of osgt1-1, we collected pollen grains from fully developed anthers and observed them with a bright field microscope. Pollen from wild-type anthers was normal: only a small fraction (nine out of 1,393) was shrunken at stage 12 (Fig. 1B; Supplemental Fig. S2). However, anthers from the heterozygote produced shrunken grains at a 49.5% frequency (915 of 1,847; Fig. 1C; Supplemental Fig. S2), indicating that OsGT1 is required for male gamete development. To determine whether those shrunken grains were caused by the T-DNA insertion, we used a Suc density gradient to separate them from normal grains (Bedinger and Edgerton, 1990; Fig. 1D). The gradient containedfour discrete bands, two lower bands that contained normal pollen with a globular shape (Fig. 1, E and F) plus two upper bands with defective grains that were shrunken or underdeveloped (Fig. 1, G and H). To see whether this defect was due to the mutation, we prepared DNA from each band and performed PCR with gene-specific primers. This resulted in amplification of the OsGT1 fragment from the two lower bands but not from the upper bands (Fig. 1I). Amplification with a gene-specific primer (R2) and a T-DNA primer (L1) did not generate bands from any of the four samples, indicating that normal pollen in the lower bands did not carry T-DNA and that abnormal pollen in the upper bands lacked genomic DNA because they were dead (Fig. 1J). | ||
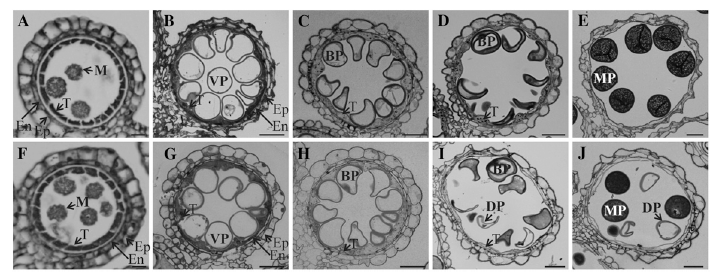
To investigate the exact time when this mutant pollen began to show the phenotypic defect, we compared wild-type and osgt1 pollen at five stages of development (Fig. 2). At the young microspore stage (stage 9), cells from the wild type were released as free microspores from the tetrad (Fig. 2A). These gradually swelled and underwent vacuolation, causing the cytoplasm and nuclei to be distributed in the periphery (Fig. 2B). Afterward, the pollen started to divide mitotically (stage 11a; Fig. 2C; Zhang et al., 2011). Until this stage, all gametophytes in the OsGT1/osgt1-1 anthers appeared normal and were identical to those of the wild type (Fig. 2, F–H). However, at stage 11b, two different types of pollen were observed from the heterozygous anthers: normal grains showing even cytosolic staining and empty pollen with a lumped staining | To investigate the exact time when this mutant pollen began to show the phenotypic defect, we compared wild-type and osgt1 pollen at five stages of development (Fig. 2). At the young microspore stage (stage 9), cells from the wild type were released as free microspores from the tetrad (Fig. 2A). These gradually swelled and underwent vacuolation, causing the cytoplasm and nuclei to be distributed in the periphery (Fig. 2B). Afterward, the pollen started to divide mitotically (stage 11a; Fig. 2C; Zhang et al., 2011). Until this stage, all gametophytes in the OsGT1/osgt1-1 anthers appeared normal and were identical to those of the wild type (Fig. 2, F–H). However, at stage 11b, two different types of pollen were observed from the heterozygous anthers: normal grains showing even cytosolic staining and empty pollen with a lumped staining | ||
| Line 14: | Line 16: | ||
We tested pollen viability with fluorescein diacetate, which stains only live cells (Heslop-Harrison and Heslop- Harrison, 1970).Whereas globular pollen emitted a bright green signal, no signal was observed from the collapsed grains from mutant plants (Supplemental Fig. S3). These | We tested pollen viability with fluorescein diacetate, which stains only live cells (Heslop-Harrison and Heslop- Harrison, 1970).Whereas globular pollen emitted a bright green signal, no signal was observed from the collapsed grains from mutant plants (Supplemental Fig. S3). These | ||
findings indicated that osgt1 pollen cannot form normal mature grains. | findings indicated that osgt1 pollen cannot form normal mature grains. | ||
| + | |||
| + | [[File:rice1.png]] | ||
| + | |||
| + | ''Figure 1. Schematic diagrams of OsGT1 and aberrant pollen phenotype in osgt1 mutant. A, T-DNA insertion positions. Start and stop codons are represented as ATG and TGA, respectively; positions of insertion are shown with triangles. Shaded boxes indicate exons; lines connecting exons are introns. Primers for genotyping and expression analysis are marked with arrows. Scale bar = 100 nucleotides. B and C, Pollen grains from wild-type (B) and OsGT1/osgt1-1 (C) anthers. Bars = 20 mm. D, Defective pollen was separated from normal grains through Suc density gradient centrifugation. Bar = 500 mm. E to H, Representative pollen from each band. Bars = 10 mm. I, PCR products by two OsGT1 primers. J, PCR products by OsGT1 primer and T-DNA primer.'' | ||
| + | |||
| + | |||
| + | |||
| + | |||
| + | [[File:table2.png]] | ||
| + | |||
| + | ''Table1.'' | ||
| + | |||
| + | |||
| + | |||
| + | [[File:table2.png]] | ||
| + | |||
| + | ''Table 2.'' | ||
| + | |||
| + | |||
| + | |||
| + | [[File:image2.png]] | ||
| + | |||
| + | ''Figure 2. Light microscopy observation of anthers at different developmental stages. Cross sections are shown from segregating wild type (A–E) and OsGT1/osgt1-1 (F–J) at early microspore stage (A and F), vacuolated stage (B and G), mitotic division stage 11a (C and H), mitotic division stage 11b (D and I), and mature pollen stage (E and J). BP, Binuclear pollen; DP, defective pollen; En, endodermis; Ep, epidermis; M, microspore; MP, mature pollen; T, tapetum; VP, vacuolated pollen. Bars = 25 mm.'' | ||
| + | |||
'''osgt1 Pollen Is Defective in Intine Formation''' | '''osgt1 Pollen Is Defective in Intine Formation''' | ||
| + | |||
To investigate the defects in osgt1 pollen further, we stained the mature grains with auramine O, which binds to exine (Dobritsa et al., 2011). This revealed that the exine was not markedly different between mutant and wild-type pollen (Fig. 3, A–D; Supplemental Fig. S4). We also monitored for the presence of intine via calcofluor white staining.Whereas defective pollen from heterozygous plants exhibited a very weak signal, wildtype pollen emitted bright blue fluorescence, demonstrating that the mutant pollen was defective in intine (Fig. 3, E–H; Supplemental Fig. S4). | To investigate the defects in osgt1 pollen further, we stained the mature grains with auramine O, which binds to exine (Dobritsa et al., 2011). This revealed that the exine was not markedly different between mutant and wild-type pollen (Fig. 3, A–D; Supplemental Fig. S4). We also monitored for the presence of intine via calcofluor white staining.Whereas defective pollen from heterozygous plants exhibited a very weak signal, wildtype pollen emitted bright blue fluorescence, demonstrating that the mutant pollen was defective in intine (Fig. 3, E–H; Supplemental Fig. S4). | ||
Staining with 496-diamidino-2-phenylindole (DAPI) showed that the defective pollen did not carry any nuclei at stage 12, whereas wild-type grains contained one vegetative nucleus and two generative nuclei (Fig. 3, I–L). Because mitotic division precedes intine construction in rice (Lu et al., 2002; Lin et al., 2009), we performed DAPI staining at stage 11, when the binuclear grains are formed. All pollen from the heterozygous plants contained two nuclei (Fig. 3, N and P), suggesting that osgt1 pollen underwent normal mitosis I. Furthermore, amido black staining showed that osgt1 grains did not accumulate cytosolic contents such as proteins (Supplemental Fig. S5), supporting our belief that the pollen abnormality developed later in the mutant. | Staining with 496-diamidino-2-phenylindole (DAPI) showed that the defective pollen did not carry any nuclei at stage 12, whereas wild-type grains contained one vegetative nucleus and two generative nuclei (Fig. 3, I–L). Because mitotic division precedes intine construction in rice (Lu et al., 2002; Lin et al., 2009), we performed DAPI staining at stage 11, when the binuclear grains are formed. All pollen from the heterozygous plants contained two nuclei (Fig. 3, N and P), suggesting that osgt1 pollen underwent normal mitosis I. Furthermore, amido black staining showed that osgt1 grains did not accumulate cytosolic contents such as proteins (Supplemental Fig. S5), supporting our belief that the pollen abnormality developed later in the mutant. | ||
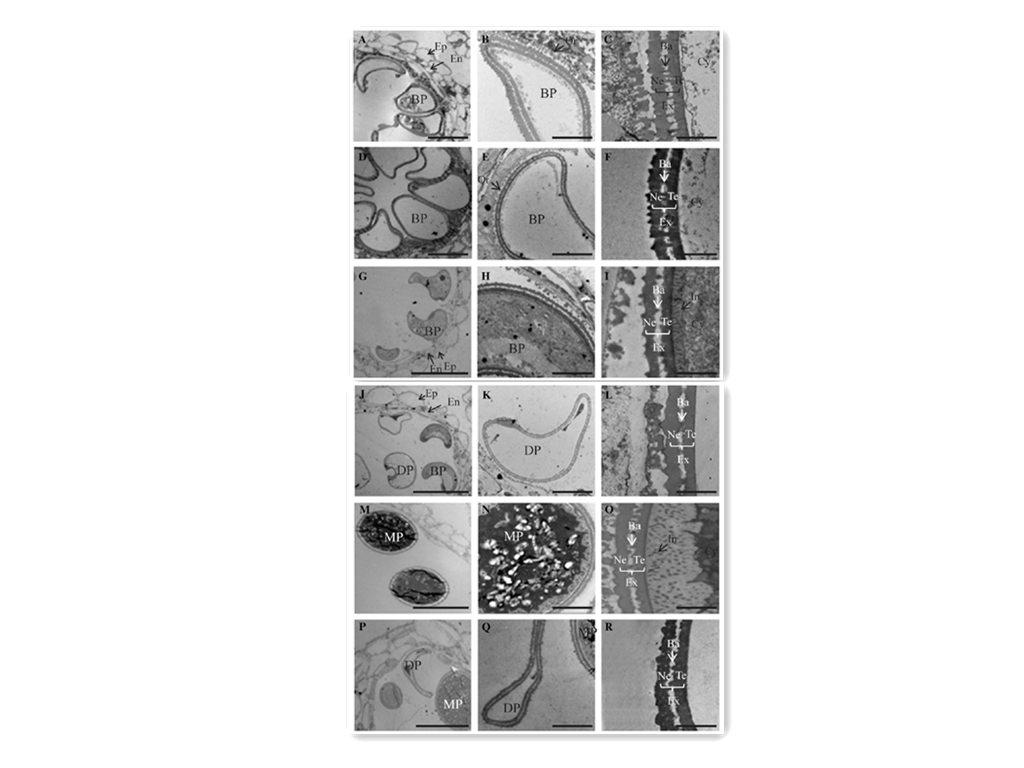
To understand the subcellular changes in osgt1 pollen, we performed transmission electron microscopy (TEM) analysis at stages 11a, 11b, and 12 (Zhang and Wilson, 2009). As revealed via bright-field microscopy, wild-type and osgt1 pollen did not differ significantly at stage 11a (Fig. 4, A–F). At the end of this stage, exine formation is generally considered complete (Lu et al., 2002; Lin et al., 2009). We noted a complete exine structure for both the wild type (Fig. 4, A–C) and osgt1 (Fig. 4, D–F) at stage 11a. However, differences were observed at stage 11b. Whereas wild-type grains had evenly stained cytoplasm (Fig. 4, G and H), mutant pollen showed severely degraded cytoplasm (Fig. 4, J and K). At higher magnification, intine accumulation was observed in wild-type pollen (Fig. 4I), but the intine was defective in osgt1 grains (Fig. 4L). At stage 12, these differences were more apparent. Whereas wildtype grains had accumulated starch granules as well as a thick intine (Fig. 4, M–O), approximately 50% (72 of 129) of the pollen from OSGT1/osgt1 anthers had an altered cytoplasm density and a severe defect in intine accumulation (Fig. 4, P–R). Because intine development precedes the accumulation of starch grains, proteins, and other inclusions in the cytosol (Lu et al., 2002; Lin et al., 2009), we speculated that OsGT1 plays a key role in the former process. | To understand the subcellular changes in osgt1 pollen, we performed transmission electron microscopy (TEM) analysis at stages 11a, 11b, and 12 (Zhang and Wilson, 2009). As revealed via bright-field microscopy, wild-type and osgt1 pollen did not differ significantly at stage 11a (Fig. 4, A–F). At the end of this stage, exine formation is generally considered complete (Lu et al., 2002; Lin et al., 2009). We noted a complete exine structure for both the wild type (Fig. 4, A–C) and osgt1 (Fig. 4, D–F) at stage 11a. However, differences were observed at stage 11b. Whereas wild-type grains had evenly stained cytoplasm (Fig. 4, G and H), mutant pollen showed severely degraded cytoplasm (Fig. 4, J and K). At higher magnification, intine accumulation was observed in wild-type pollen (Fig. 4I), but the intine was defective in osgt1 grains (Fig. 4L). At stage 12, these differences were more apparent. Whereas wildtype grains had accumulated starch granules as well as a thick intine (Fig. 4, M–O), approximately 50% (72 of 129) of the pollen from OSGT1/osgt1 anthers had an altered cytoplasm density and a severe defect in intine accumulation (Fig. 4, P–R). Because intine development precedes the accumulation of starch grains, proteins, and other inclusions in the cytosol (Lu et al., 2002; Lin et al., 2009), we speculated that OsGT1 plays a key role in the former process. | ||
| − | [[File: | + | |
| + | [[File:image3-.png]] | ||
| + | ''Figure 3. Phenotypes of pollen grains. A to D, Auramine O staining of grains from segregating wild-type (A and C) and OsGT1/ osgt1-1 (B and D) plants at stage 12 observed under bright-field (A and B) and fluorescence (C and D) microscopy. E to H, Calcofluor white staining of pollen grains from segregating wild-type (E and G) and OsGT1/osgt1-1 (F and H) plants at stage 12 observed under bright-field (E and F) and fluorescence (G and H) microscopy. I to P, DAPI staining of pollen grains from segregating wild-type (I, K, M, and O) and OsGT1/osgt1-1 (J, L, N, and P) plants at stage 12 (I–L) and stage 11 (M–P) plants observed under bright-field (I, J, M, and N) and fluorescence (K, L, O, and P) microscopy. Arrows indicate nucleus. DP, Defective pollen. Bars = 20 mm.'' | ||
| + | |||
| + | |||
| + | [[File:image4.png]] | ||
| + | |||
| + | ''Figure 4. TEM analyses of developing anthers from wild-type (A–C, G–I, and M–O) and OsGT1/osgt1-1 (D–F, J–L, and P–R) plants at stage 11a (A–F), stage 11b (G–L), and stage 12 (M–R). Ba, Bacula; BP, binuclear pollen; Cy, cytosol; DP, defective microspores; En, endothecium; Ep, epidermis; Ex, exine; In, intine; MP, mature pollen; Ne, nexine; Or, orbicule; SG, starch granule; Te, tectum. Bars = 10 mm (A, D, G, J, M, and P), 2 mm (B, E, H, K, N, and Q), and 0.5 mm (C, F, I, L, O, and R).'' | ||
| + | |||
===Expression=== | ===Expression=== | ||
Please input expression information here. | Please input expression information here. | ||
Revision as of 05:31, 4 June 2014
OsGT1 is a Golgi-localized glycosyltransferase essential for intine construction and pollen maturation, providing new insight into male reproductive development.
Contents
Annotated Information
Function
T-DNA Insertions in OsGT1 Cause a Defect in Male Gametophyte Development
We previously generated transfer DNA (T-DNA)-tagged lines of japonica rice (Jeon et al., 2000; Jeong et al., 2002) and determined flanking sequences for the insertion sites (An et al., 2003; Jeong et al., 2006). To identify genes essential for gametophyte development, we genotyped the T-DNA insertion lines; those with a segregation ratio close to 1:1:0 (wild type:heterozygote: homozygote) were selected. From 541 independentlines, we obtained eight with the segregation distortion phenotype. Here, we report detailed analyses of line 3C-00590, which carries a T-DNA insertion in the fifth intron of LOC_Os01g15780 (Fig. 1A). Because the gene encodes a protein in the glycosyltransferase family, we named it OsGT1. We found that another allele (osgt1-2) also harbors T-DNA in the fourth exon of OsGT1 (Fig.1A). This line also exhibited a 1:1:0 segregation (Table I;Supplemental Fig. S1), confirming that this phenotype is due to the T-DNA insertion to OsGT1. These segregation phenotypes suggested that the defect results from a failure in gene transfer through either the male or female gamete. To clarify the function of OsGT1 in the gametophyte, we performed reciprocal crosses between heterozygotes (OsGT1/osgt1-1) and the wild type. When OsGT1/osgt1-1 was used as a pollen receiver (female), wild-type and heterozygous progeny were obtained at a similar frequency (11:13; Table II). However, when OsGT1/osgt1-1 served as a pollen donor (male), only wild-type progeny were obtained (32:0; Table II). Reciprocal crosses between OsGT1/osgt1-2 and wild-type plants produced similar results (Table II). These indicated that mutations in OsGT1 cause defects in the male gamete.
osgt1 Grains Are Defective at the Mature Pollen Stage
To examine further the morphological defects in male gametes of osgt1-1, we collected pollen grains from fully developed anthers and observed them with a bright field microscope. Pollen from wild-type anthers was normal: only a small fraction (nine out of 1,393) was shrunken at stage 12 (Fig. 1B; Supplemental Fig. S2). However, anthers from the heterozygote produced shrunken grains at a 49.5% frequency (915 of 1,847; Fig. 1C; Supplemental Fig. S2), indicating that OsGT1 is required for male gamete development. To determine whether those shrunken grains were caused by the T-DNA insertion, we used a Suc density gradient to separate them from normal grains (Bedinger and Edgerton, 1990; Fig. 1D). The gradient containedfour discrete bands, two lower bands that contained normal pollen with a globular shape (Fig. 1, E and F) plus two upper bands with defective grains that were shrunken or underdeveloped (Fig. 1, G and H). To see whether this defect was due to the mutation, we prepared DNA from each band and performed PCR with gene-specific primers. This resulted in amplification of the OsGT1 fragment from the two lower bands but not from the upper bands (Fig. 1I). Amplification with a gene-specific primer (R2) and a T-DNA primer (L1) did not generate bands from any of the four samples, indicating that normal pollen in the lower bands did not carry T-DNA and that abnormal pollen in the upper bands lacked genomic DNA because they were dead (Fig. 1J). To investigate the exact time when this mutant pollen began to show the phenotypic defect, we compared wild-type and osgt1 pollen at five stages of development (Fig. 2). At the young microspore stage (stage 9), cells from the wild type were released as free microspores from the tetrad (Fig. 2A). These gradually swelled and underwent vacuolation, causing the cytoplasm and nuclei to be distributed in the periphery (Fig. 2B). Afterward, the pollen started to divide mitotically (stage 11a; Fig. 2C; Zhang et al., 2011). Until this stage, all gametophytes in the OsGT1/osgt1-1 anthers appeared normal and were identical to those of the wild type (Fig. 2, F–H). However, at stage 11b, two different types of pollen were observed from the heterozygous anthers: normal grains showing even cytosolic staining and empty pollen with a lumped staining pattern (Fig. 2I). At the mature stage (stage 12), those differences were even more distinct. Whereas all of the pollen from wild-type anthers was normal (Fig. 2E),one-half of the grains from anthers of heterozygotes were shrunken and exhibited exiguous cytosolic contents (Fig. 2J). We tested pollen viability with fluorescein diacetate, which stains only live cells (Heslop-Harrison and Heslop- Harrison, 1970).Whereas globular pollen emitted a bright green signal, no signal was observed from the collapsed grains from mutant plants (Supplemental Fig. S3). These findings indicated that osgt1 pollen cannot form normal mature grains.
Figure 1. Schematic diagrams of OsGT1 and aberrant pollen phenotype in osgt1 mutant. A, T-DNA insertion positions. Start and stop codons are represented as ATG and TGA, respectively; positions of insertion are shown with triangles. Shaded boxes indicate exons; lines connecting exons are introns. Primers for genotyping and expression analysis are marked with arrows. Scale bar = 100 nucleotides. B and C, Pollen grains from wild-type (B) and OsGT1/osgt1-1 (C) anthers. Bars = 20 mm. D, Defective pollen was separated from normal grains through Suc density gradient centrifugation. Bar = 500 mm. E to H, Representative pollen from each band. Bars = 10 mm. I, PCR products by two OsGT1 primers. J, PCR products by OsGT1 primer and T-DNA primer.
Table1.
Table 2.
Figure 2. Light microscopy observation of anthers at different developmental stages. Cross sections are shown from segregating wild type (A–E) and OsGT1/osgt1-1 (F–J) at early microspore stage (A and F), vacuolated stage (B and G), mitotic division stage 11a (C and H), mitotic division stage 11b (D and I), and mature pollen stage (E and J). BP, Binuclear pollen; DP, defective pollen; En, endodermis; Ep, epidermis; M, microspore; MP, mature pollen; T, tapetum; VP, vacuolated pollen. Bars = 25 mm.
osgt1 Pollen Is Defective in Intine Formation
To investigate the defects in osgt1 pollen further, we stained the mature grains with auramine O, which binds to exine (Dobritsa et al., 2011). This revealed that the exine was not markedly different between mutant and wild-type pollen (Fig. 3, A–D; Supplemental Fig. S4). We also monitored for the presence of intine via calcofluor white staining.Whereas defective pollen from heterozygous plants exhibited a very weak signal, wildtype pollen emitted bright blue fluorescence, demonstrating that the mutant pollen was defective in intine (Fig. 3, E–H; Supplemental Fig. S4). Staining with 496-diamidino-2-phenylindole (DAPI) showed that the defective pollen did not carry any nuclei at stage 12, whereas wild-type grains contained one vegetative nucleus and two generative nuclei (Fig. 3, I–L). Because mitotic division precedes intine construction in rice (Lu et al., 2002; Lin et al., 2009), we performed DAPI staining at stage 11, when the binuclear grains are formed. All pollen from the heterozygous plants contained two nuclei (Fig. 3, N and P), suggesting that osgt1 pollen underwent normal mitosis I. Furthermore, amido black staining showed that osgt1 grains did not accumulate cytosolic contents such as proteins (Supplemental Fig. S5), supporting our belief that the pollen abnormality developed later in the mutant. To understand the subcellular changes in osgt1 pollen, we performed transmission electron microscopy (TEM) analysis at stages 11a, 11b, and 12 (Zhang and Wilson, 2009). As revealed via bright-field microscopy, wild-type and osgt1 pollen did not differ significantly at stage 11a (Fig. 4, A–F). At the end of this stage, exine formation is generally considered complete (Lu et al., 2002; Lin et al., 2009). We noted a complete exine structure for both the wild type (Fig. 4, A–C) and osgt1 (Fig. 4, D–F) at stage 11a. However, differences were observed at stage 11b. Whereas wild-type grains had evenly stained cytoplasm (Fig. 4, G and H), mutant pollen showed severely degraded cytoplasm (Fig. 4, J and K). At higher magnification, intine accumulation was observed in wild-type pollen (Fig. 4I), but the intine was defective in osgt1 grains (Fig. 4L). At stage 12, these differences were more apparent. Whereas wildtype grains had accumulated starch granules as well as a thick intine (Fig. 4, M–O), approximately 50% (72 of 129) of the pollen from OSGT1/osgt1 anthers had an altered cytoplasm density and a severe defect in intine accumulation (Fig. 4, P–R). Because intine development precedes the accumulation of starch grains, proteins, and other inclusions in the cytosol (Lu et al., 2002; Lin et al., 2009), we speculated that OsGT1 plays a key role in the former process.
 Figure 3. Phenotypes of pollen grains. A to D, Auramine O staining of grains from segregating wild-type (A and C) and OsGT1/ osgt1-1 (B and D) plants at stage 12 observed under bright-field (A and B) and fluorescence (C and D) microscopy. E to H, Calcofluor white staining of pollen grains from segregating wild-type (E and G) and OsGT1/osgt1-1 (F and H) plants at stage 12 observed under bright-field (E and F) and fluorescence (G and H) microscopy. I to P, DAPI staining of pollen grains from segregating wild-type (I, K, M, and O) and OsGT1/osgt1-1 (J, L, N, and P) plants at stage 12 (I–L) and stage 11 (M–P) plants observed under bright-field (I, J, M, and N) and fluorescence (K, L, O, and P) microscopy. Arrows indicate nucleus. DP, Defective pollen. Bars = 20 mm.
Figure 3. Phenotypes of pollen grains. A to D, Auramine O staining of grains from segregating wild-type (A and C) and OsGT1/ osgt1-1 (B and D) plants at stage 12 observed under bright-field (A and B) and fluorescence (C and D) microscopy. E to H, Calcofluor white staining of pollen grains from segregating wild-type (E and G) and OsGT1/osgt1-1 (F and H) plants at stage 12 observed under bright-field (E and F) and fluorescence (G and H) microscopy. I to P, DAPI staining of pollen grains from segregating wild-type (I, K, M, and O) and OsGT1/osgt1-1 (J, L, N, and P) plants at stage 12 (I–L) and stage 11 (M–P) plants observed under bright-field (I, J, M, and N) and fluorescence (K, L, O, and P) microscopy. Arrows indicate nucleus. DP, Defective pollen. Bars = 20 mm.
Figure 4. TEM analyses of developing anthers from wild-type (A–C, G–I, and M–O) and OsGT1/osgt1-1 (D–F, J–L, and P–R) plants at stage 11a (A–F), stage 11b (G–L), and stage 12 (M–R). Ba, Bacula; BP, binuclear pollen; Cy, cytosol; DP, defective microspores; En, endothecium; Ep, epidermis; Ex, exine; In, intine; MP, mature pollen; Ne, nexine; Or, orbicule; SG, starch granule; Te, tectum. Bars = 10 mm (A, D, G, J, M, and P), 2 mm (B, E, H, K, N, and Q), and 0.5 mm (C, F, I, L, O, and R).
Expression
Please input expression information here.
Evolution
Please input evolution information here.
You can also add sub-section(s) at will.
Labs working on this gene
Please input related labs here.
References
Please input cited references here.
Structured Information
| Gene Name |
Os01g0262600 |
|---|---|
| Description |
Glycosyl transferase, group 1 domain containing protein |
| Version |
NM_001049188.1 GI:115435789 GeneID:4325125 |
| Length |
5685 bp |
| Definition |
Oryza sativa Japonica Group Os01g0262600, complete gene. |
| Source |
Oryza sativa Japonica Group ORGANISM Oryza sativa Japonica Group
Eukaryota; Viridiplantae; Streptophyta; Embryophyta; Tracheophyta;
Spermatophyta; Magnoliophyta; Liliopsida; Poales; Poaceae; BEP
clade; Ehrhartoideae; Oryzeae; Oryza.
|
| Chromosome | |
| Location |
Chromosome 1:8884740..8890424 |
| Sequence Coding Region |
8884740..8884997,8886068..8886130,8887345..8887607,8887708..8887862,8888174..8888445 |
| Expression | |
| Genome Context |
<gbrowseImage1> name=NC_008394:8884740..8890424 source=RiceChromosome01 preset=GeneLocation </gbrowseImage1> |
| Gene Structure |
<gbrowseImage2> name=NC_008394:8884740..8890424 source=RiceChromosome01 preset=GeneLocation </gbrowseImage2> |
| Coding Sequence |
<cdnaseq>atggcgaagccctcgtcggcggcggcggcgtcgggcggcggccgaggcccggcccaccaccacaggacccgcctcctcctcctgctcctcctcgccgtcgccgcctgcgcatccaccgcgggcttcctcctctgcggcgccatgctggacccctgtgacgtcgacgcgcgcaggggctcgggctcgtcggcggcggccgtcgcgaccacccgcaccggcgccgtcgcagggaaccccctcgagttcatgaggtccaagctcatgctgctcgtctcccacgagctctccctctccgggggaccattgttactgatggagttggcatttcttctgcggcaggttggttcgcaagtggtgtggataacaaaccagagatcggaagaaacaaatgatgttacatatagcttggagcataagatgttgagccatggagtgcaggtattacctgctaggggacatgaggcgattgatactgccctaaaggctgatcttgttatcttgaacactgctgttgctggcaagtggcttgatgctgtcctaaatgaccatgttccccaagttcttcccaagattttgtggtggatccatgaaatgcgagggcattactttaagcttgaatatgtcaaacatcttcctctggttgctggagccatgattgattctcatacaacggctgaatattggaagaccaggactcatgaccgtttgaaaatacagatgccacaaacttatgttgttcacctcgggaatagtaaggagttaatggaagttgctgaagataatgttgcaagaagagtcctacgggagcatatccgtgagttccttggagtacggagtgaagatcttgtgtttgcaataataaacagtgtttcacgtggaaaagggcaggacttatttcttcaagcgttttatcagggtgtccagctcatcgaacagaagaagttaaaagtgcccacaatgcatgctgtagttgtgggaagtgacataaatgctcagaccaagtttgagacacaattacgtgattttgcggtgaagaatgggattcaggaccgggttcactttgtaaacaagacattagcagtggctccttatttggctgcaactgatgtgcttgttcagaactctcaggcccgtggagaatgctttggaaggataacgattgaagcaatggcattcaagttgccagtgttgggcacggcggccggagggaccacggagatcgtcgtggacggctcaaccggcctcctgcaccccgccgggaaggaaggcgtcgcgcccctcgcgaagaacatggtaaggctcgcgagccacgaagaggacagggtctccatgggtaggaaaggctatggcagggtgaaggaaatgttcatggagcaccacatggctggtaggatcgcggccgtgttgcaggaagttctgaagaaatcgcgggaacattctcactcttga</cdnaseq> |
| Protein Sequence |
<aaseq>MAKPSSAAAASGGGRGPAHHHRTRLLLLLLLAVAACASTAGFLL CGAMLDPCDVDARRGSGSSAAAVATTRTGAVAGNPLEFMRSKLMLLVSHELSLSGGPL LLMELAFLLRQVGSQVVWITNQRSEETNDVTYSLEHKMLSHGVQVLPARGHEAIDTAL KADLVILNTAVAGKWLDAVLNDHVPQVLPKILWWIHEMRGHYFKLEYVKHLPLVAGAM IDSHTTAEYWKTRTHDRLKIQMPQTYVVHLGNSKELMEVAEDNVARRVLREHIREFLG VRSEDLVFAIINSVSRGKGQDLFLQAFYQGVQLIEQKKLKVPTMHAVVVGSDINAQTK FETQLRDFAVKNGIQDRVHFVNKTLAVAPYLAATDVLVQNSQARGECFGRITIEAMAF KLPVLGTAAGGTTEIVVDGSTGLLHPAGKEGVAPLAKNMVRLASHEEDRVSMGRKGYG RVKEMFMEHHMAGRIAAVLQEVLKKSREHSHS</aaseq> |
| Gene Sequence |
<dnaseqindica>5428..5685#4295..4357#2818..3080#2563..2717#1980..2251#710..852#1..295#atggcgaagccctcgtcggcggcggcggcgtcgggcggcggccgaggcccggcccaccaccacaggacccgcctcctcctcctgctcctcctcgccgtcgccgcctgcgcatccaccgcgggcttcctcctctgcggcgccatgctggacccctgtgacgtcgacgcgcgcaggggctcgggctcgtcggcggcggccgtcgcgaccacccgcaccggcgccgtcgcagggaaccccctcgagttcatgaggtccaagctcatgctgctcgtctcccacgagctctccctctccggtaaggagtagcagtgtgcttcagtatattctgttgaggaatcttgcgatttgttcatgttaccttttgagaatgcgatgcctccaagatcgttatatggattttatttccagtagcaaacagactgataggaaagtgaaatttctactcccagtgctcagcacgaactcatgatccctgaaaaggaaaaaaagaaaaactggaaattcggattatcacagatcacaaataaaattgtcgtatgcgtcgctctgcttgagttgtttataagcgggaaacttagtttggaagttttagcatatctacgcagttctcattgtcacagatctcatagaattgtttgctgttagtgacatctgtaactatgtctgattgtctactggtgttactggaaacacttttgttttgatgcagggggaccattgttactgatggagttggcatttcttctgcggcaggttggttcgcaagtggtgtggataacaaaccagagatcggaagaaacaaatgatgttacatatagcttggagcataagatgttgagccatggagtgcaggtatgttctacgctacttagttctgcaaattacccgtatgtcagcagaatatctaataggagttcacctcggtgaaagccaccgccatgctattcaacactaccttaaaatatggtccaggatatcatttgttcggattggaagttagatatgaaaaataacttttttttctgttcagtttgataattggactttaaagcaaagttcgatgggcaaaaatggaatttcttcaatgaatcatagtgtaaacaaaattgttgaataaatcatcttgtgggaaacaagaaattgaattaccacaatgtaatgttttagttctgaattagaacctatttaaatttttagagaatactagattgattgtttatgttgagtcacacaagaaggcacaatggaataaaatattgcacggtaagggaaaataaaagaagggtggaagttttcatataacacttgaaacttcaaggatcaaacagttcagtataaattggcattaactgtttgatccttgaaacaacatcttgtagcatgctgtgtcatgcttttgctcatgtgcattgttgagtcctaatatagaccaatatatatatatacattcattaagactaacaggatttcttgtggctggtctaatcttatcccatcattcatctgcatgttactatatgcacagaagccataatatttcttgcattgcaaatttataactgattagtatatagtagttatatactacttaagattgttgataaaccatattgatggggtgataaataaatagctacagatgaatttactctgttattattgtcttgtagtcttggagtgttagttttgagttctcttgcttgtttatttgtcagtactgctaattttgatgttctacatactcgttgcagtcctatcttggtctcatgtactatgattaccctcttaaatttgttccaaatggctgttttctcctgagataattacttaaacgtttgaaataattactacgcaatatttgatagaatatgttggagtacaaaagaaaaacaggtccaaatactttcccttcttttggcagacttaacaaacattccatgctctcgtcaattttcacactatcactgcttgcttcataggtattacctgctaggggacatgaggcgattgatactgccctaaaggctgatcttgttatcttgaacactgctgttgctggcaagtggcttgatgctgtcctaaatgaccatgttccccaagttcttcccaagattttgtggtggatccatgaaatgcgagggcattactttaagcttgaatatgtcaaacatcttcctctggttgctggagccatgattgattctcatacaacggctgaatattggaagaccaggactcatgaccgtttgaagtatgtgcaaaattattcacattctttgtaatataaatgctggatcatgtaacatttgtcaccgtttctgcttattggacttgctagtgcttttatattatttactgccctagatgatgataaacttctttggccctttgcttctctgctgaactcattggcaacttttctcttcttcaattagcctatatatgaggtttcactctataaaatgaatcatgatagctatgatcaaatgcttgttcaatgcttttaaatttcctaaatttgtacttcgttgttatataattctgatttgcttccatgtaaagaatacagatgccacaaacttatgttgttcacctcgggaatagtaaggagttaatggaagttgctgaagataatgttgcaagaagagtcctacgggagcatatccgtgagttccttggagtacggagtgaagatcttgtgtttgcaataataaacagtatgcgccatttgcagcttcttatccctgttgttattccaactatatatgtatatattgattataccaattcatttctctgattgatgttaaaatacaggtgtttcacgtggaaaagggcaggacttatttcttcaagcgttttatcagggtgtccagctcatcgaacagaagaagttaaaagtgcccacaatgcatgctgtagttgtgggaagtgacataaatgctcagaccaagtttgagacacaattacgtgattttgcggtgaagaatgggattcaggaccgggttcactttgtaaacaagacattagcagtggctccttatttggctgcaactgatgtgcttgttcagaactctcaggtgagtaataattgtaataacacttccattttctcttgttttcccagttcagtggtgtttcgtgatgcaaactgtatgccttttgtgtatttgcaattttgcatcatagtttgttatttctcttcatattctttgtgtgcgtatgtgcgcgtgcgtggaatcaatcaactgttggttaagtgagtgtgctgctagcagaaagtcaagaatttaatgtgatttacaatttttctgccattttcttttttttttgggagagttttgaagcacatctgattttaagagtaatactaactactattttttttttcatctgggaacagtaagcttgataactcgataggttgaaccactgtattatggagttaattctattattctagactattctgtcagaactattagcctgaatcctaaatagcatccaacttatcttcattgtcacaacatgcttttaagtaacttgacaggagatgttatttctacatagatagtgcagaatgcttcagtatattggcatagaatgttcagcaattgtgtttattctctacgcaatactgacaaaccattttgattactttctctagcttttgattttttacatggtctcatgctcggtcgctcacccatgaggccataaccatggttatatgtattgtttcagtgtggctaggactgcctgtaccaatcacattcatcagtaaaatatctggaactgttggtccattttttattacttgttaattgttatgccgcaacgatcagtaataatgcatatttttgcttttgtatgttccgttatggctaacaggcaataccagtctccaatttttaagaacaagacagaaaaataatattgttttcgtccaaataaaaacatatatacttgtatagatttgttattcctggatttgcctccaatttatctatggaatgtttccgtacaataataatgtctagatcaagtcaatgagcacatagttgaaagcaatgtaaaagtagggtatggtcagtctgcaaatagctgaaagggccttagctccagattggtttatgggtgtgccagttgcaaatggttctgttggcacagagccctgctcctgattaggcacgtggagaatgctgtatgttaggtatcaataagtagagctcttcaaagctagaatatccataagctgatgtgcttttgtttttgctgttcttttcgtacttaggcccgtggagaatgctttggaaggataacgattgaagcaatggcattcaagttgccagtgttggtaaaactccctccctccctctcttgaagtgctcctagttataccccattgatccacaaacacaaatagtttttgactcactagtctcgatgcaagagttgagtacaattcttttttttatcaaagtatattagaaacaaaaatatatcataattttactggggaatgcaaatccaatgacactaatttcacatgatccatttctataacctcacagttcagtggtcatgaatgattaactgcatagatctcaaggtaacacgtacgtatttatcgatggagggagtataattttctacttctattaaccatcaatctgccagctagaaagccttacgcccttaccataatatagtttggaacaaaggaattttataaatactccctccactctaaaatataaaaatataagtatttctaggattcaaagtttgtatcacaatataagcatttctgggtctggggctctatacattttcagaaatacttatatttggaatggagggaactgagggagaagttcatttcccatgggagtcaaaaaaaaaaaaggattcccacgttccaaaggagccctggaatgtgggccagtatgcctctgaatacagagttattattgacaatcatgagccttggactcaactgtggactgtctgactgtccctacaaaccctcggctcttgagcctgccatcctctgtttcagccggcatctcctgcctcatgcttccatatgacagggtgtgtttggttgcaagctacactttgccacagtttgccacgcctaaggttaggcaaatttgacaggtgtttggttgtagccacagttgtggcaagattcccctccaacaaattgggtcccacgtgtcaatggctcaaaaaagtgtggcaagattcctttaggcttagtaagttgtggctaacaatttgatcacctcaccttaggcaaggtgtggcaacttttgttggcaagtaatggtaaagtgtggctgggaaccaaacagccccacaatttcattgacttgggtttaatgtcacactgcaccctcctatatctatctatactatcagggcacggcggccggagggaccacggagatcgtcgtggacggctcaaccggcctcctgcaccccgccgggaaggaaggcgtcgcgcccctcgcgaagaacatggtaaggctcgcgagccacgaagaggacagggtctccatgggtaggaaaggctatggcagggtgaaggaaatgttcatggagcaccacatggctggtaggatcgcggccgtgttgcaggaagttctgaagaaatcgcgggaacattctcactcttga</dnaseqindica> |
| External Link(s) |